Pesticides >> Insecticides >> Cypermethrin
Cypermethrin
Cypermethrin 95%TC
Cypermethrin 25%EC
Insecticide
IRAC 3; pyrethroid
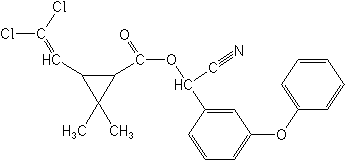
Cypermethrin NOMENCLATURE
Common name cypermethrin (BSI, E-ISO, ANSI, BAN); cyperméthrine ((f) F-ISO)
IUPAC name (RS)--cyano-3-phenoxybenzyl (1RS,3RS;1RS,3SR)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Roth: (RS)--cyano-3-phenoxybenzyl (1RS)-cis-trans-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Chemical Abstracts name cyano(3-phenoxyphenyl)methyl 3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropanecarboxylate
CAS RN [52315-07-8] (formerly [69865-47-0], [86752-99-0] and many other numbers) Development codes NRDC 149; PP383 (ICI); WL 43 467 (Shell); LE 79-600 (Rhône-Poulenc); FMC 30980 Official codes OMS 2002
Cypermethrin PHYSICAL CHEMISTRY
Composition Tech. grade is 90% pure. Mol. wt. 416.3 M.f. C22H19Cl2NO3 Form Odourless crystals; (tech., yellow-brown viscous semi-solid at ambient temperatures). M.p. 61-83 °C, depending on isomer ratio V.p. 2.0 10-4 mPa (20 ºC) KOW logP = 6.6 Henry 2.0 10-2 Pa m3 mol-1 (calc.) S.g./density 1.24 (20 ºC) Solubility In water 0.004 mg/l (pH 7). In acetone, chloroform, cyclohexanone, xylene >450, ethanol 337, hexane 103 (all in g/l, 20 ºC). Stability Relatively stable in neutral and weakly acidic media, with optimum stability at pH 4. Hydrolysed in alkaline media; DT50 1.8 d (pH 9, 25 °C); stable at pH 5 and 7 (20 °C). Relatively stable to light in field situations. Thermally stable up to 220 ºC. F.p. Not auto-flammable; non-explosive
Cypermethrin COMMERCIALISATION
History Insecticide reported by M. Elliott et al. (Pestic. Sci., 1975, 6, 537). Developed by Ciba-Geigy, ICI (both now Syngenta AG), Mitchell Cotts and Shell International Chemical Co. (now BASF AG). Patents GB 1413491 to NRDC Manufacturers Agro-Chemie; Agrochem; Aimco; Ankur; Atabay; BASF; Bharat; DE-NOCIL; Dhanuka; Dow AgroSciences; Ficom; FMC; Gharda; Jiangsu Yangnong; Krishi Rasayan; Lucava; Meghmani; Mitchell Cotts; Mitsu; Parry; Rallis; Rotam; RPG; SC Enviro Agro; Sharda; Sundat; Syngenta; Tagros; United Phosphorus
Cypermethrin APPLICATIONS
Biochemistry Acts on the nervous system of the insect, disturbs the function of neurons by interaction with the sodium channel. Mode of action Non-systemic insecticide with contact and stomach action. Also exhibits anti-feeding action. Good residual activity on treated plants. Uses Control of a wide range of insects, especially Lepidoptera, but also Coleoptera, Diptera, Hemiptera, and other classes, in fruit (including citrus), vines, vegetables, potatoes, cucurbits, lettuce, capsicums, tomatoes, cereals, maize, soya beans, cotton, coffee, cocoa, rice, pecans, oilseed rape, beet, ornamentals, forestry, etc. Control of flies and other insects in animal houses; and mosquitoes, cockroaches, houseflies and other insect pests in public health. Also used as an animal ectoparasiticide.