Products >> Fungicide >> Difenoconazole
Difenoconazole
Difenoconazole 95%TC
Difenoconazole 25%EC
Fungicide
FRAC 3; DMI: triazole
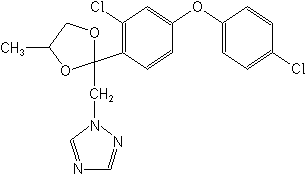
Difenoconazole NOMENCLATURE
Common name difenoconazole (BSI, draft E-ISO)
IUPAC name cis,trans-3-chloro-4-[4-methyl-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-2-yl]phenyl
4-chlorophenyl ether
Chemical Abstracts name 1-[2-[4-(4-chlorophenoxy)-2-chlorophenyl]-4-methyl-1,3-dioxolan-2-ylmethyl]-1H-1,2,4-triazole
CAS RN [119446-68-3] unstated stereochemistry Development codes
CGA 169 374 (Ciba-Geigy)
Difenoconazole APPLICATIONS
Biochemistry Sterol demethylation inhibitor. Inhibits cell membrane
ergosterol biosynthesis, stopping development of the fungus. Mode
of action Systemic fungicide with preventive and curative action.
Absorbed by the leaves, with acropetal and strong translaminar translocation.
Uses Systemic fungicide with a novel broad-range activity protecting
the yield and crop quality by foliar application or seed treatment.
Provides long-lasting preventive and curative activity against Ascomycetes,
Basidiomycetes and Deuteromycete,s including Alternaria, Ascochyta,
Cercospora, Cercosporidium, Colletotrichum, Guignardia, Mycosphaerella,
Phoma, Ramularia, Rhizoctonia, Septoria, Uncinula, Venturia spp.,
Erysiphaceae, Uredinales and several seed-borne pathogens. Used
against disease complexes in grapes, pome fruit, stone fruit, potatoes,
sugar beet, oilseed rape, banana, cereals, rice, soya beans, ornamentals
and various vegetable crops, at 30-125 g/ha. Used as a seed treatment
against a range of pathogens in wheat and barley, at 3-24 g/100
kg seed. Phytotoxicity In wheat, early foliar applications at growth
stages 29-42 might cause, in certain circumstances, chlorotic spotting
of leaves, but this has no effect on yield. Formulation types DS;
EC; FS; SC; WG. Selected tradenames: 'Dividend' (seed treatment)
(Syngenta); 'Score' (Syngenta)
Difenoconazole OTHER TRADENAMES
'Bardos' (Syngenta); 'Bogard' (Syngenta); 'Geyser' (Syngenta); 'Plandom'
(Syngenta); 'Plover' (Syngenta); 'Polyscore' (Syngenta); 'Purugen'
(Syngenta); 'Sico' (Syngenta); 'Slick' (Syngenta) mixtures: 'Arix'
(+ propiconazole) (Syngenta); 'Armure' (+ propiconazole) (Syngenta);
'Beret Gold' (+ fludioxonil) (seed treatment) (Syngenta); 'Eria'
(+ carbendazim) (Syngenta); 'Helix' (+ fludioxonil+ metalaxyl-M+
thiamethoxam) (Syngenta); 'Landor CT' (+ fludioxonil+ tebuconazole)
(seed treatment, Germany) (Syngenta); 'Neptune' (+ dinocap) (Syngenta);
'Play' (+ cyprodinil) (Syngenta); 'Spyrale' (+ fenpropidin) (Syngenta);
'Taspa' (+ propiconazole) (Syngenta); 'Trial' (+ carbendazim) (Syngenta)
Difenoconazole ANALYSIS
Residue determination by glc. Details from Syngenta.
MAMMALIAN TOXICOLOGY
Oral Acute oral LD50 for rats 1453, mice >2000 mg/kg. Skin and
eye Acute percutaneous LD50 for rabbits >2013 mg/kg. Non-irritant
to eyes and skin (rabbits). Non-sensitising to skin (guinea pigs).
Inhalation LC50 (4 h) for rats ³3300 mg/m3 air. NOEL (2 y)
for rats 1.0 mg/kg b.w. daily; (1.5 y) for mice 4.7 mg/kg b.w. daily;
(1 y) for dogs 3.4 mg/kg b.w. daily. ADI 0.01 mg/kg b.w. Other Not
teratogenic or mutagenic. Toxicity class WHO (a.i.) III EC hazard
(R22)
Difenoconazole ECOTOXICOLOGY
Birds LD50 (9-11 d) for mallard ducks >2150 mg/kg. LC50 for bobwhite
quail >4760, mallard ducks >5000 ppm. Fish LC50 (96 h) for
rainbow trout 0.8, bluegill sunfish 1.2, sheepshead minnow 0.82
mg/l. Daphnia LC50 (48 h) 0.77 mg/l. Algae EC50 (72 h) for Scenedesmus
subspicatus 1.2 mg/l. Other aquatic spp. EC50 (96 h) for Eastern
oysters 0.45 mg/l; (14 d) for Lemna gibba 18.5 mg/l. Bees Non-toxic
to honeybees; LD50 (oral) >187 mg/bee; LC50 (contact) >100
mg/bee. Worms LC50 for earthworms >610 mg/kg.
Difenoconazole ENVIRONMENTAL FATE
Animals After oral administration, difenoconazole was rapidly eliminated
practically to entirety, with urine and faeces. Residues in tissues
were not significant and there was no evidence for accumulation.
Plants Two routes of metabolism: one by a triazole route to triazolylalanine
and triazolylacetic acid; the other by hydroxylation of the phenyl
ring followed by conjugation. Soil/Environment Practically immobile
in soil, strong adsorption to soil particles, low potential to leach
below top soil layer. Soil dissipation rate is slow and dependent
on application rate. DT50 for photolysis 145 d (natural sunlight).