Pesticides >> Insecticides >> Carbaryl
Carbaryl 50%SC
Insecticide, plant growth regulator
carbamate
NOMENCLATURE
Common name carbaryl (BSI, E-ISO, (m) F-ISO, ANSI, ESA, BAN); NAC
(JMAF); sevin* (former exception, USSR)
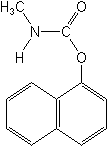
IUPAC name 1-naphthyl methylcarbamate
Chemical Abstracts name 1-naphthalenyl methylcarbamate
APPLICATIONS
Biochemistry Weak cholinesterase inhibitor. Mode of action Insecticide
with contact and stomach action, and slight systemic properties.
Uses Control of Lepidoptera, Coleoptera, and other chewing and sucking
insects, at 0.25-2.0 kg a.i./ha, on more than 120 different crops,
including vegetables, tree fruit (including citrus), mangoes, bananas,
strawberries, nuts, vines, olives, okra, cucurbits, peanuts, soya
beans, cotton, rice, tobacco, cereals, beet, maize, sorghum, alfalfa,
potatoes, ornamentals, forestry, etc. Control of earthworms in turf.
Used as a growth regulator for fruit thinning of apples. Also used
as an animal ectoparasiticide. Phytotoxicity Non-phytotoxic if used
as directed. Under certain conditions, some varieties of apple and
pear may be injured. Formulation types DP; GR; OF; RB; SC; TK; WP.
Compatibility Incompatible with alkaline materials such as Bordeaux
mixture, lime, and lime sulfur. Selected tradenames: 'Sevin' (Aventis);
'Carbamec' (PBI/Gordon); 'Efaryl' (Efthymiadis); 'Karl' (Sanonda);
'Raid' (Nagarjuna Agrichem)
OTHER TRADENAMES
'Sevimol' (Aventis); 'Sevithion' (Aventis); 'Adios' (BASF); 'Agrovin'
(AgroSan); 'Carbarex' (Crystal); 'Carbex' (Crystal); 'Panam' (Isagro,
Chemiplant); 'Slam' (BASF); 'Thinsec' (Syngenta); 'Xiweiyin' (Shenzhen
Jiangshan) mixtures: 'Sevidan' (+ endosulfan) (Aventis); 'Sevidol'
(+ gamma-HCH) (Crop Health) Discontinued names: 'Cekubaryl' * (Cequisa);
'Ravyon' * (Makhteshim-Agan); 'Savit' * (Griffin) mixtures: 'Naftil'
* (+ chlorfenson) (Pepro)
ANALYSIS
Product analysis by i.r. spectroscopy (AOAC Methods, 1995, 976.04;
CIPAC Handbook, 1970, 1, 185; 1980, 1A, 1113; FAO Specification
(CP/55)) or by hplc (G. W. Sheehan, Anal. Methods Pestic. Plant
Growth Regul., 1984, 13, 157). Residues determined by glc (ibid.,
1972, 6, 478; Man. Pestic. Residue Anal., 1987, I, 6; Anal. Methods
Residues Pestic., 1988, Part I, M2, M13; AOAC Methods, 1995, 975.40;
A. Ambrus et al., J. Assoc. Off. Anal. Chem., 1981, 64, 733), by
hplc (AOAC Methods, 1995, 985.23), by colorimetry (ibid., 964.18)
or by tlc (ibid., 968.26). For methods in drinking water, see AOAC
Methods, 1995, 991.06.
MAMMALIAN TOXICOLOGY
Reviews FAO/WHO 89 (see part 2 of the Bibliography). IARC ref. 12
class 3 Oral Acute oral LD50 for rats 264, female rats 500, rabbits
710 mg/kg. Skin and eye Acute percutaneous LD50 for rats >4000,
rabbits >2000 mg/kg. Slight eye irritant, mild skin irritant
(rabbits). Inhalation LC50 (4 h) for rats 3.28 mg/l air. NOEL (2
y) for rats 200 mg/kg diet. ADI (JMPR) 0.003 mg/kg b.w. [2000].
Toxicity class WHO (a.i.) II; EPA (formulation) I ('Tercyl' 85WP),
II ('Sevin' 80S), III EC hazard R40| Xn; R22| N; R50
ECOTOXICOLOGY
Birds Acute oral LD50 for young mallard ducks >2179, young pheasants
>2000, Japanese quail 2230, pigeons 1000-3000 mg/kg. Fish LC50
(96 h) for rainbow trout 1.3, sheepshead minnow 2.2, bluegill sunfish
10 mg/l. Daphnia LC50 (48 h) 0.006 mg/l. Algae EC50 (5 d) for Selenastrum
capricornutum 1.1 mg/l. Other aquatic spp. LC50 (96 h) for mysid
shrimp (Mysidopsis bahia) 0.0057 mg/l; LC50 (48 h) for Eastern oyster
(Crassostrea virginica) 2.7 mg/l. Bees Toxic to bees; LD50 (topical)
1 mg/bee. Worms LC50 (28 d ) 106-176 mg/kg soil. Other beneficial
spp. Toxic to beneficial insects.
ENVIRONMENTAL FATE
EHC 153 (WHO, 1994); 64 (WHO, 1986; a review of carbamate insecticides
in general). Animals In mammals, carbaryl does not accumulate in
body tissues, but is rapidly metabolised to non-toxic substances,
particularly 1-naphthol. This, together with the glucuronic acid
conjugate, is eliminated predominantly in the urine and faeces.
Metabolism of carbamate insecticides is reviewed (M. Cool &
C. K. Jankowski in "Insecticides"). Plants Metabolites
are 4-hydroxycarbaryl, 5-hydroxycarbaryl and methylol-carbaryl.
Soil/Environment Under aerobic conditions, carbaryl at 1 ppm degraded
with DT50 7-14 d in a sandy loam and 14-28 d in a clay loam.