Products >> Herbicides >> Chlorsulfuron
Chlorsulfuron
Chlorsulfuron 95%TC
Chlorsulfuron 75%WG
Chlorsulfuron 75%WP
Chlorsulfuron 25%WP
Herbicide
HRAC B WSSA 2; sulfonylurea
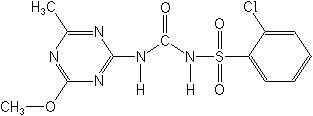
Chlorsulfuron NOMENCLATURE
Common name chlorsulfuron (BSI, draft E-ISO, (m) draft F-ISO, ANSI, WSSA)
IUPAC name 1-(2-chlorophenylsulfonyl)-3-(4-methoxy-6-methyl-1,3,5-triazin-2-yl)urea
Chemical Abstracts name 2-chloro-N-[[(4-methoxy-6-methyl-1,3,5-triazin-2-yl)amino]carbonyl]benzenesulfonamide
CAS RN [64902-72-3] EEC no. 265-268-5 Development codes DPX 4189 (DuPont); W4189
Chlorsulfuron PHYSICAL CHEMISTRY
Mol. wt. 357.8 M.f. C12H12ClN5O4S Form White crystalline solid. M.p. 170-173 °C (purity 98%) V.p. 3 10-6 mPa (25 ºC, Knudsen gas effusion) KOW logP = -0.99 (pH 7) Henry 5 10-10 (pH 5); 3.5 10-11 (pH 7); 3.2 10-12 (pH 9) (all Pa m3 mol-1, calc.) S.g./density 1.48 Solubility In water 590 (pH 5), 31800 (pH 7) (both mg/l, 25 ºC). In dichloromethane 1.4, acetone 4, methanol 15, toluene 3, hexane <0.01 (all in g/l, 25 ºC). Stability Stable to light when dry. Decomposes at 192 ºC. In aqueous solutions, DT50 23 d (pH 5, 25 ºC); >31 d (pH 7 and above). Hydrolysis is also promoted by polar organic solvents such as methanol and acetone. pKa 3.4
COMMERCIALISATION
History Herbicide reported by P. G. Jensen (Weed Control, 1980, 21st, 24). Introduced in USA in 1982, by E. I. du Pont de Nemours Co. Patents US 4127405 Manufacturers DuPont; Sharda
Chlorsulfuron APPLICATIONS
Biochemistry Branched chain amino acid synthesis (ALS or AHAS) inhibitor. Acts by inhibiting biosynthesis of the essential amino acids valine and isoleucine, hence stopping cell division and plant growth. Selectivity derives from rapid metabolism in the crop. Metabolic basis of selectivity in sulfonylureas reviewed (M. K. Koeppe & H. M. Brown, Agro-Food-Industry, 6, 9-14 (1995)). Mode of action Selective systemic herbicide, absorbed by the foliage and roots, with rapid translocation acropetally and basipetally. Uses Control of most broad-leaved weeds and some annual grasses in wheat, barley, oats, rye, triticale, flax, and on non-crop land. Applied pre-emergence, early post-emergence, pre-plant, or early post-plant incorporated, at 9-35 g/ha in crops, 140 g/ha in non-crop situations. Phytotoxicity Phytotoxic to many broad-leaved crops, particularly sugar beet and brassicas. Formulation types WG. Selected products: 'Glean' (DuPont); 'Telar' (USA) (DuPont); 'Granonet' (Agrimix); 'Lasher' (Sanonda)