Products >> Herbicides >> Chlorimuron-ethyl
Chlorimuron-ethyl 75% WDG
Herbicide
HRAC B WSSA 2; sulfonylurea
NOMENCLATURE
Common name chlorimuron-ethyl
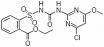
IUPAC name ethyl 2-(4-chloro-6-methoxypyrimidin-2-ylcarbamoylsulfamoyl)benzoate
Chemical Abstracts name ethyl 2-[[[[(4-chloro-6-methoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]benzoate
CAS RN [90982-32-4] Development codes DPX-F6025 (Du Pont)
chlorimuron
Common name chlorimuron (BSI, ANSI, WSSA, draft E-ISO, draft F-ISO)
IUPAC name 2-(4-chloro-6-methoxypyrimidin-2-ylcarbamoylsulfamoyl)benzoic
acid
Chemical Abstracts name 2-[[[[(4-chloro-6-methoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]benzoic
acid
CAS RN [99283-00-8]
chlorimuron
Mol. wt. 386.8 M.f. C13H11ClN4O6S
APPLICATIONS
chlorimuron-ethyl
Biochemistry Branched chain amino acid synthesis (ALS or AHAS) inhibitor.
Acts by inhibiting biosynthesis of the essential amino acids valine
and isoleucine, hence stopping cell division and plant growth. Crop
selectivity derives from plant metabolism both by homoglutathione
conjugation and by de-esterification (M. K. Koeppe & H. M. Brown,
Agro-Food-Industry, 6, 9-14 (1995)). Uses Used post-emergence for
control of important broad-leaved weeds, such as cocklebur, pigweed,
sunflower and annual morning glory, in soya beans and peanuts. Active
at 9-13 g/ha. Formulation types WG. Selected tradenames: 'Classic'
(Du Pont); 'Darban' (IPESA); 'Sponsor' (Reposo)
OTHER TRADENAMES
chlorimuron-ethyl
'Glicincas' (CAS); 'Skirmish' (FMC) mixtures: 'Canopy' (+ metribuzin)
(Du Pont); 'Concert' (+ thifensulfuron-methyl) (Du Pont); 'Preview'
(+ metribuzin) (Du Pont); 'Reliance' (+ thifensulfuron-methyl) (Du
Pont); 'Synchrony STS' (+ thifensulfuron-methyl) (Du Pont); 'Authority
Broadleaf' (+ sulfentrazone) (FMC) Discontinued names mixtures:
'Canopy XL' * (+ sulfentrazone) (Du Pont); 'Gemini' * (+ linuron)
(Du Pont); 'Lorox Plus' * (+ linuron) (Du Pont); 'New Lorox Plus'
* (+ linuron) (Du Pont); 'Spartan' * (+ sulfentrazone) (FMC)
ANALYSIS
Product analysis by hplc (R. A. Guinivan et al., Anal. Methods Pestic.
Plant Growth Regul., 1988, 16, 37). Residues determined by hplc
(J. L. Prince & R. A. Guinivan, J. Agric. Food Chem., 1988,
36, 1). Methods for sulfonylurea residues in crops, soil and water
reviewed (A. C. Barefoot et al., Proc. Br. Crop Prot. Conf. - Weeds,
1995, 2, 707).
MAMMALIAN TOXICOLOGY
chlorimuron-ethyl
Oral Acute oral LD50 for rats 4102 mg/kg. Skin and eye Acute percutaneous
LD50 for rabbits >2000 mg/kg. Not a skin irritant or eye irritant
(rabbits). Not a skin sensitiser (guinea pigs). Inhalation LC50
(4 h) for rats >5 mg/l air. NOEL (2 y) for rats 250 mg/kg diet
(12.5 mg/kg daily); (1 y) for dogs 250 mg/kg diet (6.25 mg/kg daily).
NOEL in: reproduction (2-generation) in rats 250 mg/kg diet; teratogenicity
in rats 30, rabbits 15 mg/kg. ADI 0.020 mg/kg b.w. Toxicity class
WHO (a.i.) III (Table 5); EPA (formulation) III
ECOTOXICOLOGY
chlorimuron-ethyl
Birds Acute oral LD50 (14 d) for mallard ducks >2510 mg/kg. Dietary
LC50 for mallard ducks and bobwhite quail >5620 ppm. Fish LC50
(96 h) for trout >1000, bluegill sunfish >100 mg/l. Daphnia
LC50 (48 h) 1000 mg/l. Other aquatic spp. For Lemna gibba, EbC50
0.45 ppb, ErC50 45 mg/l, EC50 (frond counts) 0.27 ppb. LC50 for
crayfish >1000 ppm. Bees LD50 (48 h) >12.5 mg/bee. Worms LC50
for earthworms (Eisenia foetida) >4050 mg/kg.
ENVIRONMENTAL FATE
chlorimuron-ethyl
Animals Chlorimuron-ethyl is rapidly and extensively metabolised
in the hen; 18 metabolites in the excreta were resolved by hplc.
Soil/Environment In soil, Kd >1.60 (pH 4.5, 5.6% o.m.), 0.28
(pH 5.8, 4.3 % o.m.), <0.03 (pH 6.5, 2.1% o.m.), <0.03 (pH
6.6, 1.1 % o.m.).