Pesticides >> Insecticides >> Tetramethrin
Tetramethrin 94%TC
Insecticide
pyrethroid
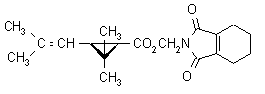
NOMENCLATURE
Common name tetramethrin (BSI, E-ISO, ANSI); phthalthrin (JMAF)
IUPAC name cyclohex-1-ene-1,2-dicarboximidomethyl (1RS,3RS;1RS,3SR)-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropanecarboxylate
Alt: cyclohex-1-ene-1,2-dicarboximidomethyl (?-cis-trans-chrysanthemate
Roth: cyclohex-1-ene-1,2-dicarboximidomethyl (1RS)-cis-trans-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropanecarboxylate
3,4,5,6-tetrahydrophthalimidomethyl (?-cis-trans-chrysanthemate
Chemical Abstracts name (1,3,4,5,6,7-hexahydro-1,3-dioxo-2H-isoindol-2-yl)methyl
2,2-dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylate
CAS RN [7696-12-0] EEC no. 213-711-6 Development codes FMC 9260;
SP 1103 (Sumitomo) Official codes OMS 1011
APPLICATIONS
Mode of action Non-systemic insecticide with contact action. Gives
rapid knockdown. Uses Normally used in combination with synergists
(e.g. piperonyl butoxide) and other insecticides for control of
flies, cockroaches, mosquitoes, wasps, and other insect pests in
public health and home and garden use. Formulation types AE; DP;
EC; EW; OE; UL; Oil. Compatibility Incompatible with mineral carriers
such as kieselguhr, acidic clays and kaolin. Selected tradenames:
'Neo-Pynamin' (Sumitomo); mixtures: 'Chinethrin' (+ permethrin+
piperonyl butoxide) (Agro-Chemie); 'Duracide' (+ piperonyl butoxide)
(Endura)
OTHER TRADENAMES
'Killgerm Py-Kill' (Killgerm) mixtures: 'Pesguard NB' (+ piperonyl
butoxide) (Sumitomo); 'Duracide P' (+ permethrin+ piperonyl butoxide)
(Endura); 'Killgerm ULV 500' (+ phenothrin [(1R)-trans- isomer])
(Killgerm); 'Lexan Forte' (+ chlorpyrifos) (Kwizda); 'Phinco - T
22' (+ permethrin+ piperonyl butoxide) (Vapco); 'Tornado-Forte+'
(+ lambda-cyhalothrin+ piperonyl butoxide) (Vapco) Discontinued
names mixtures: 'Duracide Cyp' * (+ cypermethrin+ piperonyl butoxide)
(Endura)
ANALYSIS
Product analysis of pyrethroids reviewed by E. Papadopoulou-Mourkidou
in Comp. Analyt. Profiles. By glc (M. Horiba et al., Botyu-Kagaku,
1975, 40, 123), or by u.v. spectrometry (J. Miyamoto, Anal. Methods
Pestic. Plant Growth Regul., 1973, 7, 345).
Tetramethrin: MAMMALIAN TOXICOLOGY
Reviews See A. J. Gray & D. M. Soderlund, Chapt. 5 in "Insecticides".
Oral Acute oral LD50 for rats >5000 mg/kg. Skin and eye Acute
percutaneous LD50 for rabbits >2000 mg/kg. Non-irritant to skin.
Inhalation LC50 (4 h) for rats >2.73 mg/l air. NOEL In 13 w feeding
trials, dogs receiving 5000 mg/kg diet showed no ill-effects. In
6 mo feeding trials, no-effect level for rats was 1500 mg/kg diet.
Other No evidence of oncogenicity. Toxicity class WHO (a.i.) III
(Table 5); EPA (formulation) IV
Tetramethrin: ECOTOXICOLOGY
Birds Acute oral LD50 for bobwhite quail >2250 mg/kg. Dietary
LC50 for bobwhite quail and mallard ducks >5620 ppm. Fish LC50
(96 h) for rainbow trout 3.7, bluegill sunfish 16 mg/l. Daphnia
EC50 (48 h) 0.11 mg/l. Bees Toxic to bees.
ENVIRONMENTAL FATE
EHC 98 (WHO, 1990). EHC 98 notes rapid abiotic degradation in air
and water, rapid metabolism and excretion in mammals, and no tendency
to accumulate in tissues. Animals In rats, following oral administration,
around 95% of tetramethrin (metabolised) is eliminated in the urine
and faeces within 5 days. The principal metabolite is 3-hydroxycyclohexan-1,2-dicarboximide
(J. Miyamoto et al., J. Agric. Biol. Chem., 1968, 32, 628). Soil/Environment
Degradation involves cleavage of the ester bond, leading to chrysanthemic
acid derivatives and phenoxybenzoic acid. These are further metabolised
by hydroxylation and conjugation.