Products >> Fungicides >> Fosetyl-aluminium
Fosetyl-aluminium
Fungicide
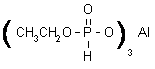
NOMENCLATURE
fosetyl-aluminium
Common namefosetyl-aluminium
Other namesefosite-Al* (rejected common name proposal); EPALCAS RN[39148-24-8]EEC no.254-320-2Development codesLS 74 783; RP 32545 (both Rhône-Poulenc)
fosetyl
Common namefosetyl (BSI, draft E-ISO, (m) draft F-ISO (since 1984)); phosethyl* ((m) draft F-ISO (before 1984))
IUPAC nameethyl hydrogen phosphonate
Chemical Abstracts nameethyl hydrogen phosphonate
Other namesefosite* (rejected common name proposal)CAS RN[15845-66-6]
PHYSICAL CHEMISTRY
fosetyl-aluminium
CompositionTech. is ³95% pure.Mol. wt.354.1M.f.C6H18AlO9P3FormColourless powder; (tech. is a white to yellowish powder).M.p.>200 °CV.p.<0.013 mPa (25 ºC)KOWlogP = -2.7 (pH 4)Henry<3.93 ´ 10-8Pa m3mol-1(calc.)SolubilityIn water 120 g/l (20 ºC). In methanol 920, acetone 13, propylene glycol 80, ethyl acetate 5, acetonitrile 5, hexane 5 (all in mg/l, 20 ºC).StabilityHydrolysis of fosetyl-aluminium occurs under extreme acid or alkaline conditions. DT505 d (pH 3), 13.4 d (pH 13). Decomposes above 200 ºC.
fosetyl
Mol. wt.110.0M.f.C2H7O3PpKa0.8
COMMERCIALISATION
HistoryFungicidal activity of aluminium salt reported by D. Horrière et al. (Phytiatr.-Phytopharm., 1977, 26, 3). The salt was introduced by Rhône-Poulenc Agrochimie (now Aventis CropScience).PatentsFR 2254276ManufacturersAventis
Fosetyl-aluminium APPLICATIONS
fosetyl-aluminium
BiochemistryActs by inhibiting germination of spores or by blocking development of mycelium and sporulation.Mode of actionSystemic fungicide, rapidly absorbed through the plant leaves or roots, with translocation both acropetally and basipetally.UsesControl of diseases caused by Phycomycetes (Phytophthora, Pythium, Plasmopara, Bremia spp., etc.) on a variety of crops including vines, fruit (citrus, pineapples, avocados, stone fruit and pome fruit), berries, vegetables, hops, ornamentals and turf. Also useful activity against several bacterial plant pathogens. Formulation typesWG; WP.CompatibilityIncompatible with foliar fertilisers.Selected tradenames:'Aliette' (Aventis); 'Fosim' (Agrimix)
Fosetyl-aluminium OTHER TRADENAMES
fosetyl-aluminium
'Prudence' (Aventis); 'R6 Triplo' (Aventis); 'Fosbel' (Probelte); 'Fosetal' (Papaeconomou); 'Manaus' (Rocca); 'Valete' (Vapco) mixtures:'Carlit' (+ benalaxyl+ mancozeb) (Aventis); 'Chipco' (+ iprodione) (Aventis); 'Mikal' (+ folpet) (Aventis); 'Rhodax' (+ mancozeb) (Aventis); 'Valiant' (+ cymoxanil+ folpet) (Aventis); 'Altigan Flash' (+ folpet) (Sipcam Phyteurop); 'Rhodax M' (+ mancozeb) (Philagro); 'Sillage' (+ metiram) (BASF)
Fosetyl-aluminium ANALYSIS
Product analysis by iodometric potentiometric titration (CIPAC Handbook, 1995, G, 82-88). Residues determined by glc of the parent compound and its metabolite. Details of methods are available from Aventis.
MAMMALIAN TOXICOLOGY
fosetyl-aluminium
OralAcute oral LD50for rats and mice >2000 mg/kg.Skin and eyeAcute percutaneous LD50for rats and rabbits >2000 mg/kg. Non-irritating to skin.InhalationLC50(4 h) for rats >1.73 mg/l air.NOEL(90 d) for rats 5000, dogs 50 000 mg/kg diet. Non-teratogenic and non-mutagenic.ADI0-3.0 mg/kg b.w.OtherNon-carcinogenic.Toxicity classWHO (a.i.) III (Table 5); EPA (formulation) III
ECOTOXICOLOGY
fosetyl-aluminium
BirdsAcute oral LD50for bobwhite quail >8000 mg/kg.FishLC50(96 h) for rainbow trout 94.3-428 mg/l.DaphniaLC50(96 h) 189 mg/l.AlgaeEC50(96 h) for Scenedesmus panonicus 21.9 mg/l.Other aquatic spp.LC50(96 h) for fiddler crab 145 mg/l.BeesNo mortality to honeybees at 0.2 mg/bee (contact).WormsHarmless.Other beneficial spp.Harmless.
ENVIRONMENTAL FATE
fosetyl-aluminium
AnimalsFosetyl-aluminium is almost completely absorbed and undergoes extensive metabolic transformation, the major end-products being CO2and phosphorous acid.PlantsThe metabolism of fosetyl-aluminium in plants proceeds through the hydrolytic cleavage of the ethyl ester bond. Phosphorous acid is detected as the major metabolite.Soil/EnvironmentIn soil, fosetyl-aluminium has an extremely short half-life under both aerobic and anaerobic conditions, with rapid dissipation and metabolism; DT50(aerobic) 20 min to 1.5 h. In water, its stability is pH-dependent.