Products >> Fungicides >> Dodine
Dodine 50%SC
Fungicide
FRAC Y; multi-site: guanidine
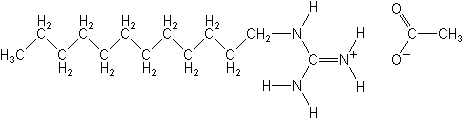
Dodine NOMENCLATURE
Common name dodine (BSI, E-ISO, (f) F-ISO, ANSI); doguadine (France);
tsitrex* (former exception, USSR); dodine acetate* (BSI, before
1969)
IUPAC name 1-dodecylguanidinium acetate
Chemical Abstracts name dodecylguanidine monoacetate
CAS RN [2439-10-3] EEC no. 219-459-5 Development codes CL 7521;
AC 5223 (both Cyanamid)
dodine free base
Common name guanidine (JMAF); dodine* (BSI, before 1969)
CAS RN [112-65-2]
Dodine APPLICATIONS
Mode of action Foliar fungicide with protective and some curative
action. Uses Dodine is used for control of scab on apples, pears,
and pecans; leaf spot diseases of cherries, olives, blackcurrants,
celery, and other crops; and foliar diseases of strawberries. Also
used on other fruit, vegetable, nut, and ornamental crops, and on
shade trees. Applied at 30-80 g a.i./100 l (0.25-1.5 kg/ha). Phytotoxicity
Peaches, plums, vines, and raspberries may be injured. Russetting
may occur on some varieties of apple (including Golden Delicious).
Formulation types SC; SL; WP. Compatibility Incompatible with anionic
surfactants, chlorobenzilate, lime sulfur, oils, oil emulsions,
and hard water. Selected tradenames: 'Cyprex' (BASF); 'Venturol'
(BASF); 'Dodene' (Sipcam); 'Dodex' (Barclay); 'Efuzin' (Agrokémia);
'Guanidol' (Caffaro); 'Melprex' (BASF, Aventis); 'Sulgen' (General
Quimica)
Dodine OTHER TRADENAMES
'Dodylon' (Papaeconomou); 'Karp-N' (Chemiplant); 'Radspor' (Truchem);
'Syllit' (Aventis, UAP)
ANALYSIS
Product analysis by titration in non-aqueous media (CIPAC Handbook,
1983, 1B, 1802; AOAC Methods, 1995, 970.07). Residues determined
by colorimetry of a complex (ibid., 964.19; Pestic. Anal. Man.,
1979, II; G. L. Sutherland, Anal. Methods Pestic., Plant Growth
Regul. Food Addit., 1964, 3, 41).
MAMMALIAN TOXICOLOGY
Reviews FAO/WHO 89, 91 (see part 2 of the Bibliography). Oral Acute
oral LD50 for male rats c. 1000 mg/kg. Skin and eye Acute percutaneous
LD50 for rabbits >1500, rats >6000 mg/kg. Irritating to skin.
NOEL In 2 y feeding trials, rats receiving 800 mg/kg diet suffered
a slight retardation of growth with no effect on reproduction or
lactation. ADI (JMPR) 0.1 mg/kg b.w. [2000]. Toxicity class WHO
(a.i.) III; EPA (formulation) I EC hazard Xn; R22| Xi; R36/38| N;
R50, R53
ECOTOXICOLOGY
Birds Acute oral LD50 for mallard ducks 1142, Japanese quail 788
mg/kg. Fish LC50 for harlequin fish (48 h) 0.53 mg/l; (96 h) 0.6
mg/l. Bees Topical LD50 for honeybees >0.011 mg/bee.
ENVIRONMENTAL FATE
Animals In rats, 95% of the applied dose is eliminated in urine
and faeces within 8 d; c. 74% of excreted material is unchanged
dodine; metabolites include creatine and guanidine derivatives.
Plants In plants, dodine is converted to creatine by the action
of a methyltransferase and simultaneous oxidative cleavage of the
dodecyl moiety.