pesticides >> Insecticides >> Alpha-Cypermethrin
Alpha-Cypermethrin
Alpha-Cypermethrin 95%TC
Alpha-Cypermethrin 10%EC
Alpha-Cypermethrin 10%SC
Alpha-Cypermethrin 10%ULV
Insecticide
pyrethroid
Alpha-Cypermethrin NOMENCLATURE
Common name alpha-cypermethrin (BSI, draft E-ISO); alpha-cypermethrine
((f) draft F-ISO)
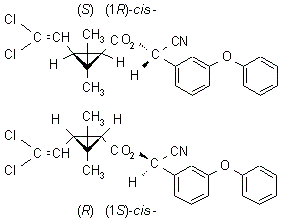
IUPAC name A racemate comprising (S)-a-cyano-3-phenoxybenzyl (1R,3R)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
and (R)-a-cyano-3-phenoxybenzyl (1S,3S)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Roth: A racemate comprising (S)-a-cyano-3-phenoxybenzyl (1R)-cis-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
and (R)-a-cyano-3-phenoxybenzyl (1S)-cis-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Chemical Abstracts name [1a(S*),3a]-(?-cyano(3-phenoxyphenyl)methyl
3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropanecarboxylate
Other names alfoxylate*; alphamethrin* (rejected common name proposals)
CAS RN [67375-30-8] correct stereochemistry; [52315-07-8] (formerly
[86752-99-0], [86753-92-6]) cypermethrin (no stereochemistry stated)
were sometimes used in Chemical Abstracts Development codes WL 85871
(Shell)
Alpha-Cypermethrin APPLICATIONS
Mode of action Non-systemic insecticide with contact and stomach
action. Acts on the central and peripheral nervous system in very
low doses. Uses Control of a wide range of chewing and sucking insects
(particularly Lepidoptera, Coleoptera, and Hemiptera) in fruit (including
citrus), vegetables, vines, cereals, maize, beet, oilseed rape,
potatoes, cotton, rice, soya beans, forestry, and other crops. Control
of cockroaches, mosquitoes, flies, and other insect pests in public
health; and flies in animal houses. Also used as an animal ectoparasiticide.
Formulation types EC; SC; TB; UL; WP. Compatibility Compatible with
most organophosphorus insecticides. Selected tradenames: 'Fastac'
(agronomic use) (BASF); 'Fendona' (public health use) (BASF); 'Renegade'
(veterinary use) (BASF); 'Alphadhan' (Dhanuka); 'Alphaguard' (Gharda);
'Bestox' (FMC); 'Bestseller' (Agriphar); 'Dominex' (FMC); 'Grander'
(Sanonda); 'Hurricane' (Nagarjuna Agrichem); 'Stop' (Biostadt)
Alpha-Cypermethrin ANALYSIS
Product analysis of pyrethroids reviewed by E. Papadopoulou-Mourkidou
in Comp. Analyt. Profiles. Residues determined by glc with ECD.
Details available from BASF.
Alpha-Cypermethrin MAMMALIAN TOXICOLOGY
Reviews JECFA 47 (see part 2 of the Bibliography). See A. J. Gray
& D. M. Soderlund, Chapt. 5 in "Insecticides". Oral
Acute oral LD50 for rats 79-400 mg/kg (in corn oil, value depending
on concentration), 474 mg tech./kg. Skin and eye Acute percutaneous
LD50 for rats and rabbits >2000 mg tech./kg; minimal irritant
to the eyes of rabbits. Inhalation LC50 (4 h) for rats 0.32 mg/l
air. NOEL In 90 d feeding trials, rats receiving 60 mg/kg diet showed
no ill-effects. ADI 0.02 mg/kg b.w. (JECFA evaluation) [1996]. Other
Non-mutagenic. Toxicity class WHO (a.i.) II; EPA (formulation) II
Alpha-Cypermethrin ECOTOXICOLOGY
Birds LD50 for quail and mallard ducks >10 000 mg/kg. Fish LC50
(96 h) for rainbow trout 0.0028 mg/l; due to rapid loss from water,
no toxic effect to fish is observed under field conditions. Daphnia
LC50 (48 h) 0.1-0.3 mg/l. Bees Toxic to bees. LD50 (24 h) 0.059
mg/bee. No toxic effect under field conditions. Worms LD50 (14 d)
for earthworms >100 mg/kg artificial soil.
Alpha-Cypermethrin ENVIRONMENTAL FATE
EHC 142 (WHO, 1992) EHC 142 notes that, although highly toxic to
fish, this is not realised under field conditions where rapid loss
from water allows recovery of affected populations. Animals See
cypermethrin. Soil/Environment Undergoes degradation in soil, DT50
c. 13 w in loamy soil.